What Does Increasing Regulatory Oversight Mean For The Pharma Supply Chain?

By developing and maintaining the ability to adapt their supply chains, companies in the pharmaceuticals sector will be well prepared to seize the opportunities ahead.
Due to more complex supply chains, policy makers worldwide are enforcing stricter regulations for manufacturing and logistics.
Selected manufacturing problems and growing public pressure have caused the US Food and Drug Administration (FDA) to strengthen GMP standards (Good Manufacturing Practices) and control them more strictly.
Within a decade, the number of FDA warning letters sent to medical device manufacturers has increased threefold, from 61 letters in 2002 to 181 in 2012 (EC A 2012).
The number of FDA inspections of drug and biologics manufacturers has also grown. In the years to come, the FDA expects domestic GMP inspections to decrease and more inspections in the foreign arena (FDA 2013) reflecting the trend towards more globalized supply chains.
The Top Ten Trends Transforming the Pharmaceutical Supply Chain
The pharmaceutical landscape continues to morph at a rapid pace, and participating companies are having to develop ever more sophisticated supply chain processes and strategies in order to deal with ongoing changes in demographics, urban migration, increasing demand for healthcare, shifts toward age related disease patterns, and a rising health awareness across the population. This paper explores the top 10 trends that are transforming the pharmaceutical supply chain. Download the White Paper if you want to learn about:
- The Rise of OTC and Generics
- The growing demand for S&OP
- Optimized execution in operations, logistics, and distribution
- Counterfeiting
- Rising healthcare costs
- Increased regulatory oversight
- Direct sales channel to the end-consumer
- Outsourcing
- Taking cues from retail and consumer goods
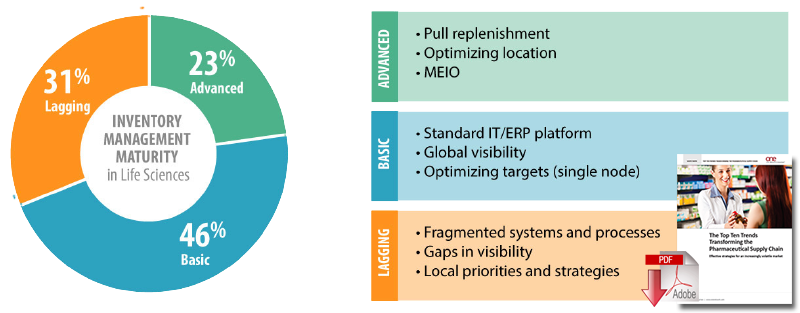
- Inventory management a top priority
At the same time, most emerging countries themselves are increasing their regulatory requirements. China published new GMP guidelines in 2012 and called on pharmaceutical companies to pursue GMP certification by the end of 2013.
Hand in hand with GMP standards, governments and international organizations such as WHO, have established and increased their requirements for Good Distribution Practices (GDP) – these standards define how life sciences products, particularly pharmaceuticals, have to be stored, transported and handled.
Higher GDP standards are looming in a number of countries – new guidelines will come into force during 2014 in Brazil, the European Union and China. Singapore is currently drafting new GDP standards (ColdChainIQPC).
Key new requirements in the European Union include risk assessment of delivery routes, temperature monitoring and reporting of temperature excursions and the use of dedicated vehicles where possible.
Challenges for the life sciences industry will have many consequences for logistics within the sector. Shifting disease patterns and innovative products will have an impact on what and how we store and transport.
Regulation, competition and cost pressure will have a direct affect logistics procedures.
Market changes and decentralized supply chains will require new and modified transportation routes.
Article Topics
One Network Enterprises News & Resources
Blue Yonder announces an agreement to acquire One Network Enterprises for $839 million Blue Yonder Acquires One Network Enterprises for $839M Companies Need to Develop New Innovative Approaches to Supply Chain Design How to Improve Cost of Goods Sold Horizontally Across the Supply Chain How the Global Pandemic Accelerated Supply Chain Visibility, Digitalization, and Automation AI and Data, the Future of Supply Chain Management AI and Supply Chain Problem Solving More One Network EnterprisesLatest in Supply Chain
Trucking Industry Pushes Back on Government’s Electric Mandates Senators Take Aim at Amazon with Warehouse Worker Protection Act Maersk Sees Silver Lining in Red Sea Shipping Challenges Happy Returns Partners With Shein and Forever 21 to Simplify Returns Baltimore Opens 45-Foot Deep Channel Following Bridge Collapse El Paso Border Delays Cost Juarez $32 Million Per Day in Economic Losses Ranking the World’s 10 Biggest Supply Chains More Supply Chain