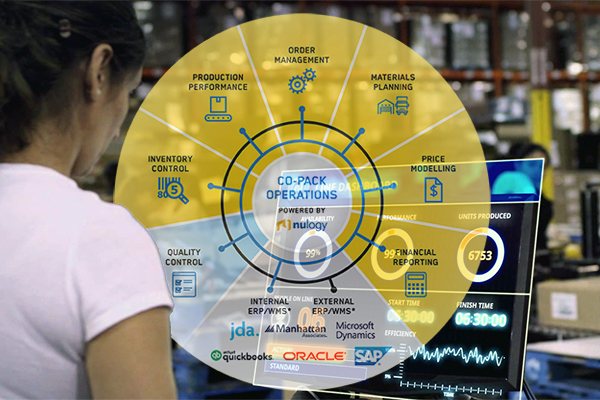
Nulogy Expands Its Agile Customization Platform for Brand Owners and Packaging Service Providers

Nulogy, a leading provider of agile supply chain solutions to consumer packaged goods (CPG) companies and their external supplier networks, announced the launch of its Enhanced Quality Solution at PACK EXPO International.
Nulogy Expands Its Agile Customization Platform with Enhanced Quality Capabilities for Effortless Compliance
With the release of Enhanced Quality, Nulogy helps brand owners and contract packaging service providers protect consumer safety with the quality control and conformance capabilities needed to uphold standards for regulated and quality-centric industries.
According to the Contract Packaging Association’s “State of the Industry Report 2018,” “70% of contract packaging and contract manufacturing companies are concerned that the regulatory environment will have a significant and possible negative impact over the next three years.”
“Supplier quality initiatives are a major priority for CPG brands, second only to cost. Our latest release ensures that we continue to support our CPG customers and their late-stage customization suppliers in upholding the high quality standards and stringent regulatory requirements that drive industries such as food, personal care, and over-the-counter products.”
“Quality and compliance is a huge part of our guarantee and we rely on best-in-class software to maintain our reputation,” said Ted Ditch, Director of Quality Assurance at AmeriPac.
“For example, the Audit Trails functionality within Nulogy’s Enhanced Quality solution has proven to be very valuable during investigations and troubleshooting, as it allows our team to see the chain of events that lead to the particular set up of a job, and follow the data all the way through to reconciliation on the line.”
Nulogy’s Enhanced Quality solution includes the following capabilities to help brand owners and their suppliers demonstrate compliance while managing costs and complexity within the late-stage customization environment:
Superior traceability
Audit trails ensure that businesses have full traceability over the modification of regulated electronic records and other high-risk activities that may impact product quality, consumer safety, or data integrity.
Evidence of safe product handling
Electronic batch records enable businesses to provide evidence that a batch of product was produced according to specification and is safe for release to market. It helps businesses fulfill one of the most critical requirements under GMP regulation. Unlike other solutions on the market which can require substantial amounts of manual entry, the automation of electronic batch recording within the Nulogy platform serves to improve data integrity and streamline processes within the production environment.
Seamless digital sign-offs
Electronic signatures make it easy for the right user to sign off on critical activities. The capability effectively controls the approval and review of regulated events through user identity verification.
Digitized and integrated quality processes
The quality inspection process is streamlined through the auto-population of production data into quality inspection forms, and is enforced through mandatory inspection checkpoints which eliminate shortcuts and workarounds.
Related White Papers
The Agile Customization Platform
Nulogy's cloud-based Agile Customization Platform allows consumer brands to respond with ease and speed to a volatile retail and consumer environment while reducing waste and costs. Download Now!
Activate Efficient & Agile Operations
Nulogy's solutions for Suppliers help you efficiently manage complex, variable workflows, increase production speed and provide enviable customer service. Download Now!
Article Topics
Nulogy News & Resources
How to Strengthen Your Position With Consumer Packaged Goods Building Strategic Partnerships Across North America Delivering a Positive Brand Experience Agile Customization Platform Operational Tools Nulogy’s Agile Customization Ecosystem Tools Navigating the Complex Regulatory Environment within Healthcare & Pharma Packaging Customization & Digitalization are Essential for Differentiated CPGs Product Experiences More NulogyLatest in Transportation
UPS Struggles in First Quarter With Steep Earnings Decline FedEx Announces Plans to Shut Down Four Facilities The Two Most Important Factors in Last-Mile Delivery Most Companies Unprepared For Supply Chain Emergency Baltimore Bridge Collapse: Impact on Freight Navigating Amazon Logistics’ Growth Shakes Up Shipping Industry in 2023 Nissan Channels Tesla With Its Latest Manufacturing Process More Transportation